Halia Rhizome
Zingiber officinale Roscoe
Zingiberaceae
DEFINITION
Halia rhizome consists of dried rhizome of Z. officinale Roscoe.
SYNONYM
Amomum zingiber L., Zingiber blancoi Massk [ 1 , 2 ].
VERNACULAR NAMES
Halia (Malay), sheng jiang (Chinese), ingi, chukku (Tamil), ginger (English) [ 3 , 4 ].
CHARACTER
The dried rhizome is yellowish brown with characteristic aromatic odour and pungent aromatic taste.
IDENTIFICATION
Plant Morphology
A soft, upright perennial herb, up to 1.50 m high. Stem non-woody, covered with leaf sheath, with a subterranean, digitately branched rhizome. Sheathing leaves linear lanceolate, about 5-30 cm long and 8-20 mm wide, dark green, alternate, smooth on the upper surface and slightly rough on the underneath which is covered with very fine hairs; ligule light green in color. Flower stems shorter than leaf stems, bearing a few flowers, each surrounded by a thin bract and situated in axils of large, greenish yellow obtuse bracts, closely arranged at end of flower stem in whorl forming collectively an ovate-oblong spike; each flower shows a superior tubular calyx, split part way down one side; corolla orange yellow, composed of a tube, divided into 3 separated blunt lobes, linear oblong, ca. 2-2.5 cm long, slightly longer than bractea; dorsal lobe broader, 12 mm by 10 mm; 6 staminodes in 2 rows, the outer row of 3 inserted at mouth of corolla; the posterior 2, small, horn-like; the anterior petaloid, purple and spotted and divided into 3 rounded lobes; an inferior, 3-celled ovary with tufted stigma. Fruit a capsule with small arillate seeds [ 1 , 5 , 6 ]. Ginger rhizome occurs in horizontal, laterally flattened, irregularly branching pieces, 3-16 cm long, 3-4 cm wide, up to 2 cm thick, sometimes split longitudinally, pale yellowish buff or light brown externally, longitudinally striated, somewhat fibrous; branches known as “fingers” arise obliquely from the rhizomes, flattish, obovate, short, about 1-3 cm long; fracture, short and starchy with projecting fibres. Internally, yellowish brown, showing a yellow endodermis separating the narrow cortex from the wide stele, and numerous scattered fibrovascular bundles; abundant scattered oleoresin cells with yellow contents and numerous larger greyish points; vascular bundles, scattered on the whole surface [ 1 , 7 , 8 , 9 , 10 ]; aromatic smell, slightly hot in taste [ 11 ].
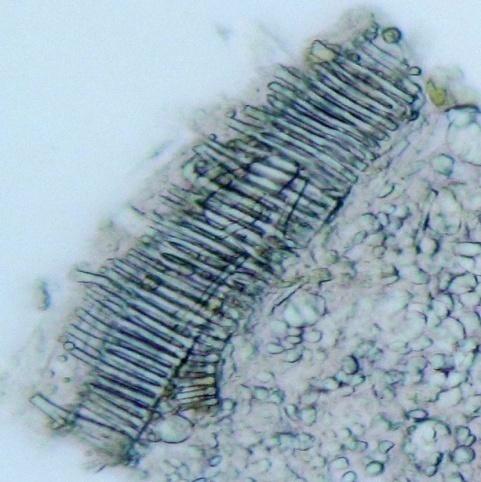
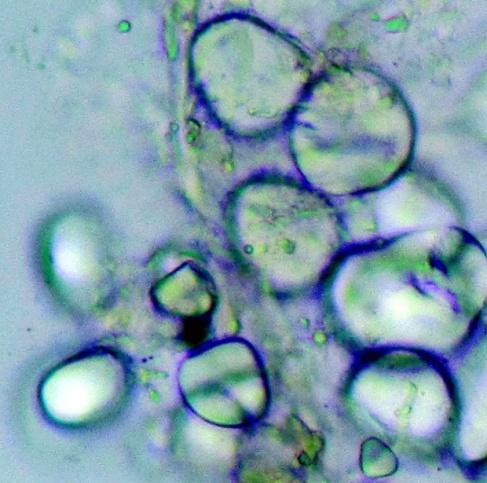
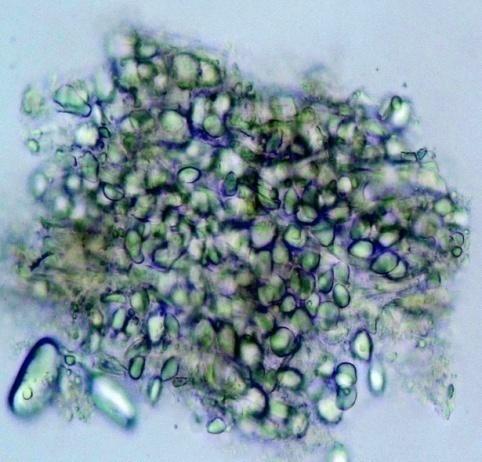
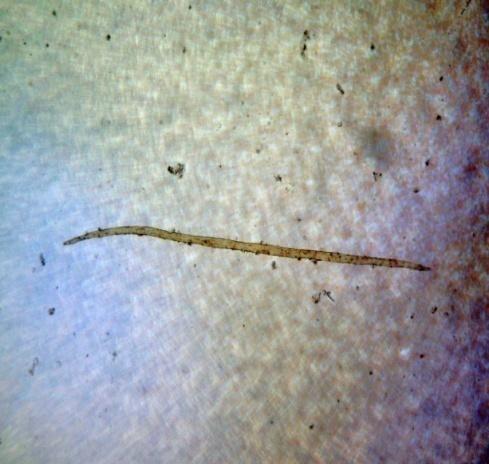
Microscopy
The rhizome powder has numerous starch granules, almost all are simple, of various sizes from round to oval, oblong to sub-rectangular. The thin-walled septate fibers are found in groups and sometimes found associated with the vessels. The reticulate thickened vessels frequently showing regularly arranged rectangular pits and the spirally thickened vessels are much smaller and very few. The abundant parenchyma cells are composed of very thin-walled cells, rounded to oval with small intercellular spaces [ 3 , 4 ].
Colour Tests
Observed colour of solution after treatment with various reagents:
| H2SO4 (conc.) | Dark brown to black |
| NaOH (5%) | Yellow to light brown |
| KOH (5%) | Yellow to light brown |
Thin Layer Chromatography (TLC)
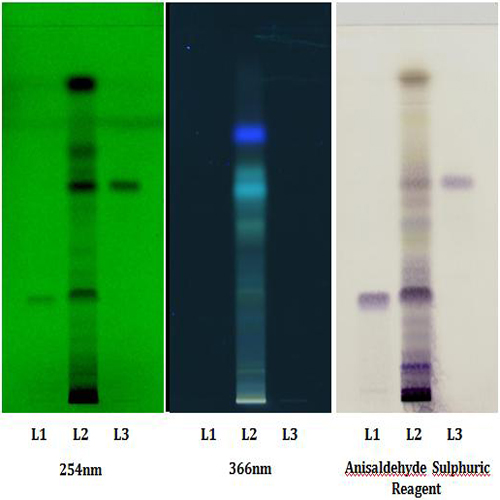
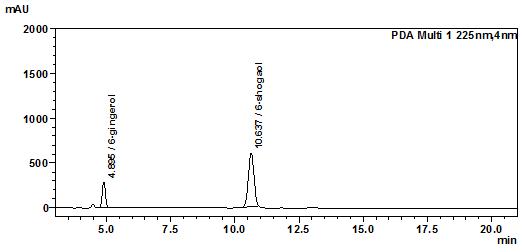
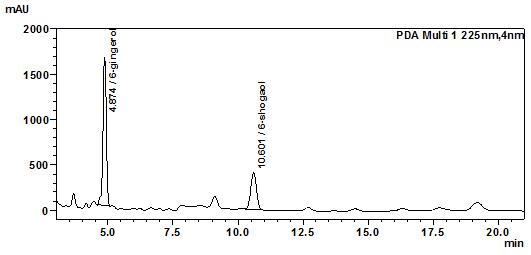
Figure 3 : TLC profiles of a mixture of 6-shogaol and 6-gingerol standards (S) and methanol extract of Z. officinale rhizome (L) after spraying with anisaldehyde-sulphuric acid reagent and observed under (a) visible light.
| Test Solutions | Weigh about 0.5 g of Z. officinale dried rhizome powder in a 50 mL screw-capped conical flask and add 10 mL methanol. Sonicate in a water bath of 60°C for 30 min. Cool the mixture and allow the insoluble matter to settle. Filter the mixture and use the filtrate as the test solution. |
| Standard solution | Separately dissolve 5.0 mg of 6-gingerol and 6-shogaol standards in 10 mL of methanol to give 500 µg/mL solutions. Pipette 1 mL of 6-gingerol and 1 mL of 6-shogaol into a 5-mL volumetric flask and make up to volume with methanol to give 100 µg/mL solution. |
| Stationary Phase | HPTLC silica gel 60 F254, 5 x 10 cm |
| Mobile phase | Toluene-ethyl acetate, 3:1 (v/v) |
| Application |
|
| Development distance | 8 cm |
| Drying | Air drying |
| Detection |
|
High Performance Liquid Chromatography (HPLC)
| Test solution | Extract about 1.0 g of Z. officinale dried rhizome powder with 20 mL of ethanol by reflux method at a temperature of 60°C for 30 min. Filter the mixture through a filter paper. Evaporate the filtrate to dryness using a rotary evaporator. Then, dissolve the dried extract in 10 mL of methanol. Sonicate the mixture for 10 min. Filter the solution through a 0.45 µm syringe filter and inject the filtrate into the HPLC column. |
| Standard solution | Separately dissolve 5.0 mg of 6-gingerol and 6-shogaol standards in 5 mL of methanol to give 1000 µg/mL stock solutions. Sonicate the solutions for 15 min. Mix the standard solutions to produce a 100 µg/mL solution. |
| Chromatographic system |
Detector: UV 225 nm Column: C18 (5 µm, 4.6 mm I.D x 150 mm) Column oven temperature: 30°C Flow rate: 1.0 mL/min Injection volume: 20 µL |
| Mobile Phase (Isocratic mode) |
Isocratic elution using the mobile phase described below:
|
| System suitability requirement |
Perform at least five replicate injections of the standard mixture (100 µg/mL). The requirements of the system suitability parameters are as follow:
|
| Acceptance criteria |
|
Table 1 : The Relative Retention Time (RRT) for the four characteristic peaks
| Standard | RRT |
| 6-gingerol | 0.46 |
| 6-shogaol (as reference) | 1.00 |
Note: The RRTs provided only serve as a guidance
PURITY TESTS
| Foreign Matter |
| Not more than 2% |
| Ash Contents | |
| Total ash | Not more than 8% |
| Acid-insoluble ash | Not more than 2% |
| Loss on Drying |
| Not more than 10% |
| Extractive Values | |
| Water-soluble extracts | |
| Hot method | Not less than 22% |
| Cold method | Not less than 17% |
| Ethanol-soluble extracts | |
| Hot method | Not less than 9% |
| Cold method | Not less than 6% |
SAFETY TEST
| Heavy Metals | |
| Arsenic | Not more than 5.0 mg/kg |
| Mercury | Not more than 0.5 mg/kg |
| Lead | Not more than 10.0 mg/kg |
| Cadmium | Not more than 0.3 mg/kg |
| Microbial Limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
CHEMICAL CONSTITUENTS
The aqueous extract of Z. officinale rhizome has been found to contain hexanoic phosphate and gingerlanosterol [ 12 ].
The ethanolic extracts had diarylheptanoids (e.g. (3S,5S)-3,5-diacetoxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptane, 3,5-diacetoxy-1-(3-methoxy-4,5-dihydroxy-phenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, (3R,5S)-3-acetoxy-5-hydroxy-1, 7-bis(4-hydroxy-3-methoxyphenyl)heptane, (3R,5S)-3,5-dihydroxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, (5S)-5-acetoxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptan-3-one, 5-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptan-3-one, 5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-7-(3,4-dihydroxy-5-methoxyphenyl)heptan-3-one, 1,5-epoxy-3-hydroxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl) heptane and 1,5-epoxy-3-hydroxy-1-(3,4-hydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane), acyclic diarylheptanes (e.g. (3R,5S)-3,5-di-acetoxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptane, (3R,5S)-3,5-diacetoxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, (3S,5S)-3,5-dihydroxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptane, (3R,5S)-3,5-dihydroxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptane), diarylheptanones (e.g. 5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)-7-(3,4-dihydroxyphenyl)heptan-3-one, 7-(3,4-dihydroxy-5-methoxyphenyl)-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)heptan-3-one, 5-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptan-3-one, hexahydrocurcumin, 1,7-bis(4-hydroxy-3-methoxyphenyl)heptane-3,5-dione), cyclic diarylheptanes (e.g. 3-acetoxy-1,5-epoxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, 1,5-epoxy-3-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(3,4-dihydroxyphenyl)heptane), diarylheptenones (e.g. 1,7-bis(4-hydroxy-3-methoxyphenyl)hept-4-en-3-one, gingerenone C), paradols (e.g. [6]-paradol, [8]-paradol, zingerone), gingerdiols (e.g. (3S,5S)-[6]-gingerdiol, (3R,5S)-[6]-gingerdiol, (3R,5S)-3,5-diacetoxy-[6]-gingerdiol), gingerols (e.g. [4]-gingerol, [6]-gingerol, [8]-gingerol, [10]-gingerol, 5-acetoxy-[6]-gingerol, [6]-gingeroldiacetate), gingerdione (e.g. [10]-gingerdione), dehydrogingerdiones (e.g. 1-dehydro-[6]-gingerdione, 1-dehydro-[8]-gingerdione), shogaols (e.g. [6]-shogaol, [10]-shogaol, [6]-dehydroshogaol, 6-hydroxy-[6]-shogaol), steroids (e.g. ß-sitosterol, stigmasterol, 6ß-hydroxystigmast-4-en-3-one) and others (e.g. 1-(3-methoxy-4-hydroxy-phenyl)-propan-1,2-diol, 1,7-bis(4-hydroxy-3-methoxyphenyl)heptane-3,5-diol, 6-(hydroxymethyl)tetrahydro-2H-pyran-3,4-5-triol, 5-[4-hydroxy-6-(4-hydroxyphenethyl)tetrahydro-2H-pyran-2-yl]-3-methoxybenzene-1,2-diol, 1-(3,4-dimethoxyphenyl)-5-hydroxy-decan-3-one, 4a,5ß-dihydroxybisabola-2,10-diene, galanolactone, citrylidenmalonsaeure, shogasulfonic acid A, 6-gingesulfonic acid, (E)-geranylferulic acid, (Z)-geranylferulic acid, (2R,3R,4S,5S,6R)-2((1S,2S,4S)-4-hydroxy-2-methylcyclohexyloxy)-sodium 6-gingesulfonate, sodium (E)-7-hydroxy-1,7-bis(4-hydroxyphenyl)hept-5-ene-3S-sulfonate, sodium (E)-7-hydroxy-1,7-bis(4-hydroxyphenyl)hept-5-ene-3R-sulfonate, hydroxyl-cineole-10-O-ß-D-glucosidase, trans-ß-sesquiphellandrol, trans-sesquipiperitol) [ 13 , 14 , 15 , 16 , 17 ].
The methanol extract has been reported to have gingerols (e.g. [6]-gingerol, [8]-gingerol, [10]-gingerol), gingerdiones (e.g. dehydro-[6]-gingerdione, dehydro-[10]-gingerdione), shogaols (e.g. [6]-shogaol, [8]-shogaol, [10]-shogaol), zingerines (e.g. [6]-zingerine, [8]-zingerine, [10]-zingerine), [6]-gingediol and [6]-paradol [ 18 , 19 , 20 ]. Whereas the methanolic aqueous extract had [6]-gingerol, [8]-gingerol, [10]-gingerol, [6]-shogaol, [6]-paradol, [6]-gingeacetate, [4]-gingesulfonic acid, [6]-gingesulfonic acid and shogasulfonic acids A-D [ 21 ].
The chloroform extract had [6]-gingerol, [10]-gingerol, [6]-shogaol, [10]-shogaol and hexahydrocurcumin [ 22 ].
The dichloromethane extract had diarylheptanoids (e.g. (Z)-1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-4-hepten-3-one, (E)-1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-4-hepten-3-one, 1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-5-methoxyheptan-3-one, 1-(4’-hydroxy-3’,5’-dimethoxyphenyl)-7-(4’-hydroxy-3’-methoxyphenyl)-4-hepten-3-one, 1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-3-hydroxy-5-acetoxyheptane, 1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-3,5-heptadione, meso and (3S,5S)-3,5-diacetoxy-1,7-bis-(4’-hydroxy-3’-methoxyphenyl)heptane, 3-acetoxy-1,5-epoxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, epimer of 3-acetoxy-1,5-epoxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane at C-3, 1,5-epoxy-3-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, epimer of 1,5-epoxy-3-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane at C-3, 1,5-epoxy-3-hydroxy-1-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane), paradols (e.g. [1]-paradol, [6]-paradol, [7]-paradol, [8]-paradol, [9]-paradol, [10]-paradol, [11]-paradol, [13]-paradol, methyl [6]-paradol, methyl [8]-paradol, dihydro-[6]-paradol, acetoxydihydro-[6]-paradol, methyl ether derivative of acetoxydihydro-[6]-paradol, 1-hydroxy-[6]-paradol), gingerols (e.g. [4]-gingerol, [5]-gingerol, [6]-gingerol, [7]-gingerol, [8]-gingerol, [10]-gingerol, methyl [4]-gingerol, methyl [6]-gingerol, [4]-isogingerol, methyl [6]-isogingerol, acetoxy-[4]-gingerol, acetoxy-[6]-gingerol, acetoxy-[8]-gingerol, acetoxy-[10]-gingerol, methyl acetoxy-[6]-gingerol, methoxy-[4]-gingerol, methoxy-[6]-gingerol, methoxy-[8]-gingerol, methoxy-[10]-gingerol, demethoxy-[6]-gingerol, 5-acetoxy-3-deoxy-[6]-gingerol, 1-dehydro-[6]-gingerol, 6-dehydro-[6]-gingerol), shogaols (e.g. (E)-[4]-shogaol, (E)-[5]-shogaol, (Z)-[6]-shogaol, (E)-[6]-shogaol, (Z)-[8]-shogaol, (E)-[8]-shogaol, (Z)-[10]-shogaol, (E)-[10]-shogaol, (E)-[12]-shogaol, methyl (E)-[4]-shogaol, methyl (E)-[6]-shogaol, methyl (E)-[8]-shogaol, [6]-isoshogaol, demethoxy-[6]-shogaol, 3-acetoxy-3-dihydrodemethoxy-[6]-shogaol, 6-hydroxy-[6]-shogaol, 6-hydroxy-[8]-shogaol, 6-hydroxy-[10]-shogaol, 3-dihydro-[6]-demethoxyshogaol), gingerdiones (e.g. [6]-gingerdione, [8]-gingerdione, [10]-gingerdione, [12]-gingerdione, 1-dehydro-[3]-gingerdione, 1-dehydro-[6]-gingerdione, 1-dehydro-[8]-gingerdione, 1-dehydro-[10]-gingerdione, 1-dehydro-3-dihydro-[10]-gingerdione), gingerdioles (e.g. [4]-gingerdiol, [6]-gingerdiol, [8]-gingerdiol, [10]-gingerdiol, 5-acetoxy-[4]-gingerdiol, 3-acetoxy-[4]-gingerdiol, 5-acetoxy-[6]-gingerdiol, stereoisomer of 5-acetoxy-[6]-gingerdiol, 5-acetoxy-[7]-gingerdiol, methyl 5-acetoxy-[4]-gingerdiol, methyl 5-acetoxy-[6]-gingerdiol, diacetoxy-[4]-gingerdiol, diacetoxy-[6]-gingerdiol, diacetoxy-[8]-gingerdiol, methyl diacetoxy-[4]-gingerdiol, methyl diacetoxy-[6]-gingerdiol, methyl diacetoxy-[8]-gingerdiol, methyl diacetoxy-[10]-gingerdiol, (2E)-geranial acetal of [4]-gingerdiol, (2Z)-neral acetal of [6]-gingerdiol, (2E)-geranial acetal of [6]-gingerdiol, acetaldehyde acetal of [6]-gingerdiol, cyclic methyl orthoester [6]-gingerdiol, cyclic methyl orthoester [10]-gingerdiol) and others (e.g. 6-(4’-hydroxy-3’-methoxyphenyl)-2-nonyl-2-hydroxytetrahydropyran, 1-(4’-hydroxy-3’-methoxyphenyl)-2-nonadecen-1-one, 1-(3’,4’-dimethoxyphenyl)-2-nonadecen-1-one, 1-(4’-hydroxy-3’-methoxyphenyl)-7-decen-3-one, 1-(4-hydroxy-3-methoxyphenyl)-2,4-dehydro-6-decanone, 1-(4’-hydroxy-3’-methoxyphenyl)-7-octen-3-one, 1-(4’-Hydroxy-3’-methoxyphenyl)-7-dodecen-3-one, 3-(4’-hydroxyphenyl)-1-propanal, 3-(4’-hydroxy-3’-methoxyphenyl)-1-propanal, 3-(3’,4’-dihydroxy-5’-methoxyphenyl)-1-propanal, 3-(4’-hydroxy-3’,5’-dimethoxyphenyl)-1-propanal, 3-(3’-hydroxy-4’,5’-dimethoxyphenyl)-1-propanal, 2-dehydro-3-(3’,4’-dimethoxyphenyl)-1-propanal, 2-dehydro-3-(3’,4’,5’-trimethoxyphenyl)-1-propanal, 4-(4’-hydroxyphenyl)-2-butanone, 4-(3’,4’-dihydroxy-5’-methoxyphenyl)-2-butanone, 4-(4’-hydroxy-3’,5’-dimethoxyphenyl)-2-butanone, 4-(3’-hydroxy-4’,5’-dimethoxyphenyl)-2-butanone, 4-(3’,4’,5’-trimethoxyphenyl)-2-butanone, 5-(4’-hydroxy-3’-methoxyphenyl)-pent-2-en-1-al, 5-(4’-hydroxy-3’-methoxyphenyl)-3-hydroxy-1-pentanal, 1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-4-heptene-3-one, 1,7-bis-(4’-hydroxy-3’-methoxyphenyl)-3,5-heptadione, p-hydroxybenzaldehyde, vanillin, 4-vinylguaiacol, 4-vinylpyrogallol monomethyl ether, 4-vinylsyringol, acetovanillone, methyl vanillate, 2-(4’hydroxy-3’-methoxyphenyl)ethyl acetate, ferulic acid, zingerol, zingerone, zingerone methyl ether) [ 23 , 24 , 25 ].
The petroleum ether extract had [4]-gingerol, [6]-dehydrogingerdione and [6]-dihydrogingerdione [ 26 ].
Essential oils of the rhizome composed of allo aroma dendrene, a-bergamotene, ß-besabolene, endo borneol, endo bornyl acetate, ß-bourbonene, ?-cadinene, calamenene, calarene, camphene, camphor, trans-2-caren-4-ol, 1,8-ceneole, z-citral (neral), citral (geranial), citronella, ß-citronellol, citronellyl acetate, a-copaene, ß-cubebene, cuparene, a-curcumene, cyclosativen, 1-decanol, ß-elemene, ?-elemene, elemol, (-)-epiglobulol, ß-eudesmol, ß-farnasene, farnesene, (-)-farnesol, trans-geraniol, geranyl acetate, germacrene B, a-guaen, 2-heptanol, juniper camphor, ledol, linalool, 6-methyl-5-hepten-2-one, a-muurolene, ß-myrcene, myrtenal, nerolidol, nerol, 2-nonanone, cis-ocimene, a-phellandrene, ß-phellandrene, a-pinene, 2-ß-pinene, a-selinene, ß-selinene, epibicyclo sesquiphellandrene, ß-sesquiphellandrene, cis-ß-sesquiphellandrol, trans-ß-sesquiphellandrol, terpinene-4-ol, a-terpineol, a-terpinolene, torreyol, 2-undecanone, verbenone, viridiflorol and zingiberene [ 27 , 28 , 29 ].
The rhizome has also been reported to contain diarylheptanoids (e.g. 5-hydroxy-7-(4-hydroxyphenyl)-1-(4-hydroxy-3-methoxyphenyl)-3-heptanone, 3,5-diacetoxy-7-(3,4-dihydroxyphenyl)-1-(4-hydroxy-3-methoxyphenyl)heptane, 5-hydroxy-7-(4-hydroxy-3,5-dimethoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)-3-heptanone, 5-hydroxy-7-(4-hydroxy-3,5-dimethoxyphenyl)-1-(4-hydroxy-3-methoxyphenyl)-3-heptanone, (3R,5S)-3,5-dihydroxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptane, (3S,5S)-3,5-diacetoxy-1,7-bis(3,4-dihydroxyphenyl)heptane, (4E,6E)-7-(3,4-dihydroxy-5-methoxyphenyl)-1-(4-hydroxy-3-methoxyphenyl)hepta-4,6-dien-3-one, 5-hydroxy-1,7-bis(4-hydroxy-3-methoxyphenyl)heptan-3-one, 7-(3,4-dihydroxyphenyl)-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)heptan-3-one, (E)-1,7-bis(4-hydroxy-3-methoxyphenyl)hept-1-ene-3,5-dione, (E)-7-(4-hydroxy-3-methoxyphenyl)-1-(4-hydroxyphenyl)-hept-1-ene-3,5-dione, 1,5-epoxy-3-hydroxy-1-(3,4-dihydroxy-5-methoxyphenyl)-7-(4-hydroxy-3-methoxyphenyl)heptane, (3S,5S)-3,5-diacetoxy-1,7-bis(3,4-dihydroxyphenyl)heptane, curcumin, tetrahydrocurcumin, hexahydrocurcumin), diarylheptenones (e.g. gingerenone A-C, isogingerenone B), gingerdiols (e.g. [6]-gingerdiol, (3R,5S)-5-acetoxy-3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)decane, (3R,5S)-3-acetoxy-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)decane, (3R,5S)-3,5-diacetoxy-1-(4-hydroxy-3-methoxyphenyl)decane and (3R,5S)-3,5-diacetoxy-1-(3,4-dimethoxyphenyl)decane) and [10]-dehydrogingerdione [ 30 , 31 , 32 , 33 , 34 ].
On the other hand, the crude rhizome had ginger proteases (GP-I and GP-II), 1,5-epoxy-3-hydroxy-1-(3-methoxy-4,5-dihydroxyphenyl)-7-(4-hydroxyphenyl)-heptane, 10-O-ß-D-glucopyranosyl-hydroxy cineole, 1-(4-O-ß-D-glucopyranosyl-3-methoxyphenyl)-3,5-dihydroxydecane, 5-O-ß-D-glucopyranosyl-3-hydroxy-1-(4-hydroxy-3-methoxyphenyl)decane, [6]-gingerol, [8]-gingerol, [10]-gingerol, [6]-shogaol, [8]-shogaol, [10]-shogaol, [6]-dehydroshogaol, [8]-dehydroshogaol, [10]-dehydroshogaol and [1]-dehydrogingerdione [ 35 , 36 , 37 , 38 , 39 , 40 ].
MEDICINAL USES
Uses described in folk medicine, not supported by experimental or clinical data
Traditionally taken orally to stimulate carminative effect and relief indigestion. It also consumed as a decoction for stomach ache and women after childbirth. It may be applied on the skin for rubefacient and counterirritant purpose [ 41 ].
Biological and pharmacological activities supported by experimental data
Antidiarrheal activity
Methanol extract of Z. officinale rhizome (up to 100 µg/mL) inhibited Escherichia coli heat-labile enterotoxin (LT)-induced diarrhea by blocking the binding of LTB to cell-surface receptor ganglioside GM1 with an IC50 value of 2.0 µg/mL using GM1-enzyme-linked immunosorbent assay [ 42 ].
Antihyperlipidimic acitivity
Ethanol extract of Z. officinale rhizome (200 mg/kg/day) administered orally to adult male albino rabbits for a duration of 10 weeks significantly (P<0.01) decreased serum triglycerides, serum phospholipids, low-density lipoprotein cholesterol, serum and tissue cholesterol levels and significantly increased high-density lipoprotein cholesterol [ 43 ].
Ethanol extract of Z. officinale rhizome (200 mg/kg/day) administered orally to streptozotocin-induced diabetic male and female Wistar rats for a duration of 20 days significantly (p<0.01) decreased serum total cholesterol, triglycerides, liver and pancreas thiobarbituric acid reactive substances values and significantly (p<0.01) increased the level of high-density lipoprotein [ 44 ].
Aqueous extract of Z. officinale rhizome (500 mg/kg/day) administered orally and intraperitoneally to adult female Sprague Dawley rats for a duration of 4 weeks significantly (p<0.05) decreased cholesterol level with no changes in triglycerides level [ 45 ].
Antioxidant activity
Extract of Z. officinale (100 mg/kg/day) administered orally to lead acetate-induced apoptosis male Wistar rats for a duration of 10 weeks significantly (p<0.05) increased plasma superoxides dismutase activity, plasma catalase activity and decreased the plasma malondialdehyde [ 46 ]. Antithrombotic activity Aqueous extract of Z. officinale rhizome (500 mg/kg/day) administered orally to adult female Sprague Dawley rats for a duration of 4 weeks significantly (p<0.05) decreased (50%) the synthesis of thromboxane B [ 45 ].
Aqueous extract of Z. officinale rhizome (500 mg/kg/day) administered intraperitoneally to adult female Sprague Dawley rats for a duration of 4 weeks showed no reduction of thromboxane B synthesis [ 45 ]. Pro-fertility activity Aqueous extract of Z. officinale rhizome (24 mg/mL) administered orally three times weekly to metiram induced-testicular damage of sexually mature male albino mice for a duration of 6 weeks significantly (p<0.05) increased the diameter of seminiferous tubules and epithelial height of testis, increased spermatogenic cells and decreased the percentage of Bax positive germ cells [ 47 ].
Aqueous extract of Z. officinale rhizome (500 and 1000 mg/kg/day) administered orally to adult male rats for a duration of 14 and 28 days significantly (p<0.05) increased weight of testis and epididymis, sperm count and motility, testosterone level and significantly (p<0.05) decreased malonhydialdehyde level [ 48 ].
Antidiabetic activity
Aqueous extract of Z. officinale root (500 mg/kg/day) administered orally to alloxan-induced diabetic albino rats for a duration of 6 weeks significantly (p<0.05) decreased serum glucose level [ 49 ].
The juice of Z. officinale rhizome (4mL/kg/day) administered orally to streptozotocin-induced diabetic male Sprague Dawley rats for a duration of 6 weeks significantly (p<0.05) decreased fasting blood glucose level and significantly (p<0.05) increased insulin level [ 50 ].
Antifungal activity
Ethanol extract of Z. officinale rhizome (10 mg/mL) inhibited the growth of Candida albicans with MIC values of 2 mg/mL by using antifungal disk assay [ 51 ].
Clinical studies
A clinical trial to study the antiemetic property of Z. officinale as compared to metoclopramide and placebo for day case surgery was carried out. It is a prospective, randomised double-blind controlled trial which involved 120 gynaecological patients of American Society of Anesthesiologists graded 1-3; aged over 16 years old, scheduled for elective laparoscopic surgery on a day stay basis. The patients were divided into 3 groups, they were given two capsules containing 10 mg metoclopramide, 1 g powdered Z. officinale rhizome, and 1 g lactose for three respective patient groups, at 1 hour before anaesthesia induction. The observations were made at discharge from the recovery ward, at hospital discharge and 24 hours postoperatively. Results showed a decreased in postoperative nausea or vomiting incidence. The requirement of postoperative antiemetic agents were also decreased as compared to the placebo treated patients [ 53 ].
A clinical trial to compare ginger and vitamin B6 for the treatment of nausea and vomiting in pregnancy was conducted. It is a randomised double-blind controlled trial which involved 70 pregnant women with nausea at or before 17 weeks gestation. The patients took 2 capsules containing powdered of Z. officinale root (1 g/day) or vitamin B6 (40 mg/day) after breakfast and dinner for 4 days. Z. officinale was found to be significantly more effective in alleviating the severity of nausea as compared to vitamin B6 group. Z. officinale also was equally effective with vitamin B6 group in reducing the number of vomiting episode during early pregnancy period [ 54 ].
SAFETY INFORMATION
Preclinical studies (Toxicology studies)
Acute toxicity
A patented standardized ethanol extract of Z. officinale rhizome EV.EXT™ 33 (up to 100 mg/kg) administered orally to male Wistar rats for duration of 3 hours showed no changes in systolic blood glucose or heart rate [ 52 ].
Oral single dose acute toxicity study on female Sprague Dawley rats (aged between 8 and 12 weeks old) using aqueous mixture of Z. officinale rhizome powder on the parameters observed which includes behaviors, body weight, food and water intakes. All rats were observed for 14 days prior to necropsy. No death was found throughout the study period. Necropsy revealed no significant abnormality. No-observed-adverse-effect level (NOAEL) is more than 2,000 mg/kg body weight [ 55 ].
Z. officinale root (2500 mg/ kg) in saline administered orally as a single dose to adult male albino rats for a duration of 24 hours significantly (P<0.0001) decreased blood pressures and heart rate and induced prenecrotic changes in cardiac tissues with LD50 value of > 2500 mg/kg [ 56 ]. Methanol and aqueous extract of Z. officinale root (up to 17.5 g/kg bw) administered orally as a single dose to male Swiss mice (8-10 weeks old) for a duration of 48 hours showed no toxic effect with LD50 value of 10.25 and 11.75 g/kg bw respectively [ 57 ].
Sub-acute toxicity
Dried Z. officinale roots (500 mg/kg/day) in saline administered orally to adult male albino rats for a duration of 28 days showed hypotension and bradycardia effect with degenerative changes in cardiac myocyte fibers [ 56 ]. Fine powder of Z. officinale roots (up to 2000 mg/kg/day) in gum arabic administered orally to male and female Sprague Dawley rats for a duration of 35 days showed no toxic effect in behavior, growth, and food and water consumption [ 58 ].
Teratogenicity
Z. officinale rhizome tea (up to 50 g/L/day) administered orally to pregnant Sprague Dawley rats (8-12 weeks old) for a duration of 5 days from gestation day 6 to gestation day 15 showed no teratogenic effect with advanced skeletal development of fetuses and increased growth of surviving fetuses [ 59 ].
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Precaution
Z. officinale rhizome is not recommended for children less than 6 years of age [ 3 ]. Contact dermatitis on the fingers has been found in sensitive patients [ 60 ].
DOSAGE
For motion sickness: 0.5 g, 2–4 times daily in adults and children more than 6 years [ 61 ].
For dyspepsia: 2–4 g daily, as powdered plant material or extracts [ 61 ].
STORAGE
Store below 30°C. Protect from light and moisture.
REFERENCES
- Standard of Asean herbal medicine. Jakarta, Indonesia: ASEAN countries. 1993.
- Bisset NG. Max Wichtl’s herbal drugs & phytopharmaceuticals. Boca Raton, FL, CRC Press. 1994.
- WHO. WHO monographs on selected medicinal plants. Vol. 1. Geneva: WHO. 1999;p.277-287
- The Ayurvedic pharmacopoeia of India, Vol. II, Part – 1. New Delhi, Government of India Ministry of Health and Family Welfare Department of Indian Systems of Medicine and Homeopathy. 1999.
- Keys JD. Chinese herbs, their botany, chemistry and pharmacodynamics. Rutland, VT, CE Tuttle. 1976.
- Youngken HW. Textbook of pharmacognosy, 6th ed. Philadelphia, Blakiston. 1950.
- Pharmacopoeia of the People’s Republic of China (English ed.). Guangzhou, Guangdong Science and Technology Press. 1992.
- British pharmacopoeia. London, Her Majesty’s Stationery Office. 1993.
- African pharmacopoeia, Vol. 1. 1st ed. Lagos, Organization of African Unity, Scientific, Technical & Research Commission. 1985.
- The Japanese pharmacopoeia XIII. Tokyo, Ministry of Health and Welfare. 1996.
- Zingiber officinale in selected medicinal plants of India (a monograph of identity, safety and clinical usage), CHEMEXIL. 1992;p.362.
- Chung IM, Ali M, Ahmad A. Isolation and characterization of water-soluble new gingerlanosterol from Zingiber officinale rhizomes. Asian Journal of Chemistry. 2005;17(3):1915-1920.
- Imm J, Zhang G, Chan L-K, Nitteranon V, Parkin KL. [6]-Dehydroshogaol, a minor component in ginger rhizome, exhibits quinone reductase inducing and anti-inflammatory activities that rival those of curcumin. Food Research International. 2010;43:2208-2213.
- Ma J, Jin X, Yang L, Liu Z-L. Diarylheptanoids from the rhizomes of Zingiber officinale. Phytochemistry. 2004;65:1137-1143.
- Peng F, Tao Q, Wu X, Dou H, Spencer S, Mang C, Xu L, Sun L, Zhao Y, Li H, Zeng S, Liu G, Hao X. Cytotoxic, cytoprotective and antioxidant effects of isolated phenolic compounds from fresh ginger. Fitoterapia. 2012;83:568-585.
- Li F, Wang Y, Parkin KL, Nitteranon V, Liang J, Yang W, Li Y, Zhang G, Hu Q. Isolation of quinone reductase (QR) inducing agents from ginger rhizome and their in vitro anti-inflammatory activity. Food Research International. 2011;44:1597-1603.
- Hong SS, Oh JS. Phenylpropanoid ester from Zingiber officinale and their inhibitory effects on the production of nitric oxide. Archives of Pharmacal Research. 2012;35(2):315-320.
- He XG, Bernart MW, Lian LZ, Lin LZ. High-performance liquid chromatography–electrospray mass spectrometric analysis of pungent constituents of ginger. Journal of Chromatography A. 1998;796:327-334.
- Araya JJ, Zhang H, Prisinzano TE, Mitscher LA, Timmermann BN. Identification of unprecedented purine-containing compounds, the zingerines, from ginger rhizomes (Zingiber officinale Roscoe) using a phase-trafficking approach. Phytochemistry. 2011;72:935-941.
- Lee SW, Lim JH, Kim MS, Jeong JH, Song GY, Lee WS, Rho MC. Phenolic compounds isolated from Zingiber officinale roots inhibit cell adhesion. Food Chemistry. 2011;128:778-782.
- Hori Y, Miura T, Hirai Y, Fukumura M, Nemoto Y, Toriizuka K, Ida Y. Pharmacognostic studies on ginger and related drugs -part 1: five sulfonated compounds from Zingiberis rhizome (Shokyo). Phytochemistry. 2003;62:613-617.
- Lin RJ, Chen CY, Chung LY, Yen CM. Larvicidal activities of ginger (Zingiber officinale) against Angiostrongylus cantonensis. Acta Tropica. 2010;115:69-76.
- Kikuzaki H, Nakatan N. Cyclic diarylheptanoids from rhizomes of Zingiber officinale. Phytochemistry. 1996;43(1):273-277.
- Jolad SD, Lantz RC, Chen GJ, Bates RB, Timmermann BN. Commercially processed dry ginger (Zingiber officinale): composition and effects on LPS-stimulated PGE2 production. Phytochemistry. 2005;66:1614-1635.
- Jolad SD, Lantz RC, Solyom AM, Chen GJ, Bates RB, Timmermann BN. Fresh organically grown ginger (Zingiber officinale): composition and effects on LPS-induced PGE2 production. Phytochemistry. 2004;65:1937-1954.
- Rahuman AA, Gopalakrishnan G, Venkatesan P, Geetha K, Bagavan A. Mosquito larvicidal activity of isolated compounds from the rhizome of Zingiber officinale. Phytotherapy Research. 2008;22(8):1035-1039.
- Kizhakkayil J, Sasikumar B. Characterization of ginger (Zingiber officinale Rosc.) germplasm based on volatile and non-volatile components. African Journal of Biotechnology. 2012;11(4):777-786.
- Bednarczyk AA, Galetto WG, Kramer A. cis- and trans-ß-sesquiphellandrol. Two new sesquiterpene alcohols from oil of ginger (Zingiber officinale Roscoe). Journal of Agricultural and Food Chemistry. 1975;23(3):499-501.
- Smith RM, Robinson JM. The essential oil of ginger from Fiji. Phytochemistry. 1981;20(2):203-206.
- Kikuzaki H, Kobayashi M, Nakatani N. Diarylheptanoids from rhizomes of Zingiber officinale. Phytochemistry. 1991;30(11):3647-3651.
- Kikuzaki H, Tsai SM, Nakatani N. Gingerdiol related compounds from the rhizomes of Zingiber officinale. Phytochemistry. 1992;31(5):1783-1786.
- Endo K, Kanno E, Oshima Y. Structures of antifungal diarylheptenones, gingerenones A, B, C and isogingerenone B, isolated from the rhizomes of Zingiber officinale. Phytochemistry. 1990;29(3):797-799.
- Li N, Wang L, Zu L, Wang K, Di L, Wang Z. Antioxidant and cytotoxic diarylheptanoids Isolated from Zingiber officinale rhizomes. Chinese Journal of Chemistry. 2012;30(6):1351-1355.
- Choi SY, Park GS, Lee SY, Kim JY, Kim YK. The conformation and CETP inhibitory activity of [10]-dehydrogingerdione isolated from Zingiber officinale. Archives Pharmacal Research. 2011;34(5):727-731.
- Choi KH, Laursen RA. Amino-acid sequence and glycan structures of cysteine proteases with proline specificity from ginger rhizome Zingiber officinale. European Journal of Biochemistry. 2000;267:1516-1526.
- Zhao Y, Tao QF, Zhang RP, Zhou CX, Dou H, Shi SY, Xiao YC, Sun LL, Zeng S, Huang KX, Zhang XD, Li XK. Two new compounds from Zingiber officinale. Chinese Chemistry Letters. 2007;18:1247-1249.
- Sekiwa Y, Kubota K, Kobayashi A. Isolation of novel glucosides related to gingerdiol from ginger and their antioxidative activities. Journal of Agricultural and Food Chemistry. 2000:48(2):373–377.
- Kizhakkayil J, Sasikumar B. Characterization of ginger (Zingiber officinale Rosc.) germplasm based on volatile and non-volatile components. African Journal of Biotechnology. 2012;11(4):777-786.
- Wu TS, Wu YC, Wu PL, Chern CY, Leu YL, Chan YY. Structure and synthesis of [n]-dehydroshogaols from Zingiber officinale. Phytochemistry. 1998;48(5):889-891.
- Charles R, Garg SN, Kumar S. New gingerdione from the rhizomes of Zingiber officinale. Fitoterapia. 2000;71:716-718.
- Burkil IH. A dictionary of the economic products of the Malay Peninsula. Kuala Lumpur: Ministry of Agriculture Malaysia. 1935;p.2300-2301.
- Chen JC, Huang LJ, Wu SL, Kuo SC, Ho TY, Hsiang CY. Ginger and its bioactive component inhibit enterotoxigenic Escherichia coli heat-labile enterotoxin-induced diarrhea in mice. Journal of Agricultural and Food Chemistry. 2007;55:8390-8397.
- Bhandari U, Sharma JN, Zafar R. The protective action of ethanolic ginger (Zingiber officinale) extract in cholesterol fed rabbits. Journal of Ethnopharmacology. 1998;61:167-171.
- Bhandari U, Kanojia B, Pillai KK. Effect of ethanolic extract of Zingiber officinale on dyslipidaemia in diabetic rats. Journal of Ethnopharmacology. 2005;97:227-230.
- Thomson M, Al-Qattan KK, Al-Sawan SM, Alnaqeeb MA, Khan I, Ali M. The use of ginger (Zingiber officinale Rosc.) as a potential anti-inflammatory and antithrombotic agent. Prostaglandins, Leukotrienes and Essential Fatty Acids. 2002;67(6):475-478.
- Khaki AA, Khaki A. Antioxidant effect of ginger to prevents lead-induced liver tissue apoptosis in rat. Journal of Medicinal Plants Research. 2010;4(14):1492-1495.
- Sakr SA, Badawy GM. Effect of ginger (Zingiber officinale R.) on metiram-inhibited spermatogenesis and induced apoptosis in albino mice. Journal of Applied Pharmaceutical Science. 2011;1(4):131-136.
- Morakinyo AO, Adeniyi OS, Arikawe AP. Effects of Zingiber officinale on reproductive functions in the male rat. African Journal of Biomedical Research. 2008;11:329-334.
- Jafri SA, Abass S, Qasim M. Hypoglycemic effect of ginger (Zingiber officinale) in alloxan induced diabetic rats (Rattus norvegicus). Pakistan Veterinary Journal. 2011;31(2):160-162.
- Akhani S, Vishwakarma S, Goyal R. Antidiabetic activity of Zingiber officinale in streptozotocin-induced type I diabetic rats. Journal of Pharmaceutical and Pharmacology. 2004;56:101-105.
- Atai Z, Atapour M, Mohseni M. Inhibitory effect of ginger extract on Candida albicans. American Journal of Applied Sciences. 2009;6(6):1067-1069.
- Weidner MS, Sigwart K. The safety of a ginger extract in the rat. Journal of Ethnopharmacology. 2000;73:513–520.
- Phillips S, Ruggier R, Hutchinson SE. Zingiber officinale (ginger)-an antiemetic for day case surgery. Anaesthesia. 1993;48:715-717.
- Jenabi E, Mohammad-Alizadeh CS. Comparing ginger and vitamin B6 for the treatment of nausea and vomiting in pregnancy: a randomized controlled trial. Midwifery. 2009;25:649–653.
- Teh BP, Hamzah NF, Rosli SNS, Yahaya MAF, Zakiah I, Murizal Z. Acute oral toxicity study of selected Malaysian medicinal herbs on Sprague Dawley rats. Institute for Medical Research, Ministry of Health; 2012. Report No.: HMRC 11-045/01/ZO/RH/K.
- Iman A. Elkhishin, Ibrahim A. Awwad. A study of the cardiovascular toxic effects of Zingiber officinale (ginger) in adult male albino rats and its possible mechanisms of action. Mansoura Journal of Forensic Medicine and Clinical Toxicology. 2009;17:109-125.
- Shalaby MA, Hamowieh AR. Safety and efficacy of Zingiber officinale roots on fertility of male diabetic rats. Food and Chemical Toxicology. 2010;48:2920–2924.
- Rong X, Peng G, Suzuki T, Yang Q, Yamahara J, Li Y. A 35-day gavage safety assessment of ginger in rats. Regulatory Toxicology and Pharmacology. 2009;54:118–123.
- Jenny MW. Effect of ginger tea on the fetal development of Sprague-Dawley rats. Reproductive Toxicology. 2000;14:507–512.
- Seetharam KA, Pasricha JS. Condiments and contact dermatitis of the finger tips. Indian Journal of Dermatology, Venereology and Leprology. 1987;53:325–328.
- Blumenthal M, Busse WR, Goldberg A, Gruenwald J, Hall T, Klein S, Riggins CW, Rister RS. The complete German commission e monographs, therapeutic guide to herbal medicines (Zingiberis rhizoma). American Botanical Council, Austin, Texas. 1998;p.135-136.